Introduction
Ectopic pregnancy (EP) refers to blastocyst implantation outside the uterine cavity. The incidence of EP is rising, which can be explained by the increased use of assisted reproductive technologies, tubal surgeries, and improved diagnostic tools [1]. EP is medically important because it is associated with reduced fertility and an increased risk of subsequent EPs [2]. Treatment options for EP involve surgical and medical intervention [1]. Medical treatment primarily uses a regimen of methotrexate (MTX) in cases of early, unruptured EP, and is reported to be safe and effective [1].
MTX is a folic acid analog that competitively binds to dihydrofolic acid reductase, an enzyme that converts dihydrofolate to tetrahydrofolate. This binding prevents the reduction of folate to its active form, tetrahydrofolate. It transports one carbon group during the synthesis of purine nucleotides and thymidylates. MTX impairs DNA synthesis, DNA repair, and cellular replication [3]. MTX selectively acts on rapidly dividing cells, such as trophoblast cells, which comprise the implantation site during early gestation [4]. MTX enters the liver via the hepatic artery and is rapidly cleared from the body by the kidneys, with 90% of the intravenous dose excreted unchanged within 24 h of injection [5,6].
Two common regimens are available for MTX: multidose [MTX 1.0 mg/kg intramuscularly (i.m.) daily; days 0, 2, 4, and 6 alternated with folinic acid 0.1 mg/kg orally on days 1, 3, 5, and 7]; and single dose (MTX 0.4 to 1.0 mg/kg or 50 mg/m2 i.m. without folinic acid) [7]. Current regimens of MTX for the treatment of EP reduce the total MTX dose that patients receive compared to other indications such as chemotherapy. Therefore, the incidence of therapy-related side effects was low [8]. Most side effects of MTX are minor, such as nausea, vomiting, stomatitis, and diarrhea, and have rarely been reported to be linked to pancreatitis.
Acute pancreatitis (AP) is a common disease that affects the digestive system. AP is diagnosed on the basis on the fulfillment of 2 out of 3 of the following criteria : clinical (upper abdominal pain), laboratory (serum amylase or lipase >3× upper limit of normal) and/or imaging (CT, MRI, ultrasonography) criteria [9]. Various etiologies of AP, most of which are due to gallstones. Other common causes of pancreatitis are alcohol consumption, hyperlipidemia, hypercalcemia, sphincter of Oddi dysfunction, drugs and toxins, which may also be idiopathic or traumatic [10]. The management of AP involves providing active treatment, including fluid resuscitation, nutritional support, analgesics, and etiological treatment [11]. At present, three cases of MTX-induced pancreatitis have been reported [12,13]. We report a case of a 40-year-old woman who underwent MTX therapy for an unruptured EP and developed AP.
Case
A 40-year-old gravida 2, para 1 woman visited the emergency department with upper abdominal pain. She had nausea and a low-grade fever, with a maximum temperature of 37.8°C. Abdominal examination revealed severe tenderness in the epigastric area, and both the right and left upper quadrants without muscle guarding or rebound tenderness.
The patient did not consume any other medications or alcohol. She had visited another hospital four weeks ago because of lower abdominal pain and was diagnosed with a left tubal EP. On that day, her total serum hCG level was 23,544 mIU/mL. She had been treated with a single dose of MTX (99.1 mg), intramuscularly four weeks before her visit to this hospital. A week after MTX injection, her total serum hCG level was 13,166 mIU/mL, and 4,620 mIU/mL after two weeks. Three weeks after the injection the total serum hCG level was 1,400 mIU/mL, and transvaginal sonography revealed a decrease in the size of the left adnexal mass.
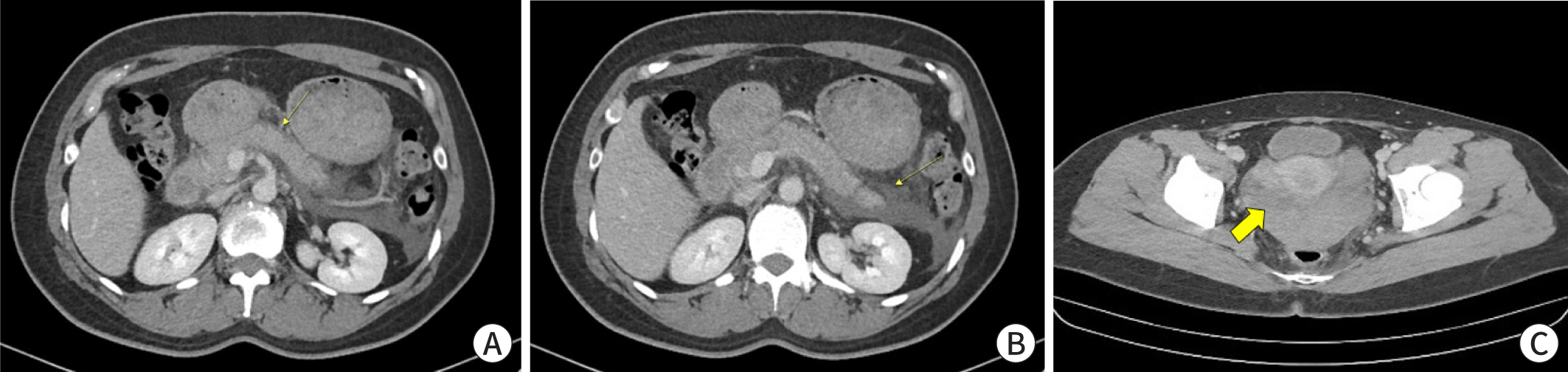
Abdominal and pelvic computed tomography scan showed diffuse acute interstitial edematous pancreatitis with peripancreatic fluid collection and infiltration (Fig. 1A) and acute hematoma in the cul-de-sac without active bleeding (Fig. 1C). Upper abdominal ultrasonography findings were consistent with that of AP (Fig. 2A, 2B).

Transvaginal ultrasonography revealed an empty uterus with 2.53 mm thick endometrium and a probable ectopic mass at the left adnexa, measuring 3.15 cm in the largest diameter, with degenerated gestational sac inside, but no fetal pole (Fig. 3A). A hematoma sized 7.9×3.5 cm2 was noted in the posterior cul-de-sac (Fig. 3B).

According to the laboratory investigations, hemoglobin was 12.6 g/dL (reference range, 12.0–16.0 g/dL), WBC was 8,400 μL (4,000–10,000 μL), platelet counts were 349,000 μL (150,000–450,000 μL), total bilirubin was 0.38 mg/dL (≤1.2 mg/dL) with direct bilirubin level of 0.14 mg/dL (≤0.25 mg/dL), AST was 15 IU/L (≤33 IU/L), ALT was 8 IU/L (≤33 IU/L), ALP was 87 IU/L (30–120 IU/L), total cholesterol was 193 mg/dL (≤200 mg/dL), which were within the normal range. The enzymes showed elevated levels: LDH 282 IU/L (135–214 IU/L), amylase 1,864 IU/L (28–100 IU/L), lipase 4,394 IU/L (13–60 IU/L), CRP 0.97 mg/dL (≤0.5 mg/dL), triglyceride 178 mg/dL (28–150 mg/dL), fibrinogen 458.1 mg/dL (180–350 mg/dL), FDP 13.7 μg/mL (≤5 μg/mL), D-dimer 5.44 mg/L (≤0.59 mg/L). HCG was detected on urine examination, and the total hCG level on admission was 1,289.0 mIU/mL. Considering her clinical manifestations, laboratory tests, CT scan and abdominal ultrasound findings, the diagnosis of AP was confirmed.
The patient was admitted to the hospital and received conservative medical management with analgesia, hydration and fasting. She received gabexate 20 mg intravenously daily for five days and 100,000 units injection units of ulinastatin intravenously per daily for two days. Those gabexate mesylate and ulinastatin are protease inhibitors, which are accepted as a potential treatment to inhibit the pancreatic inflammation in AP [14].
She was administered ciprofloxacin 400 mg every 12 h intravenously for five days during her hospitalization.
On fourth day after hospitalization, repeated measurements of her amylase and lipase levels were within normal limits: amylase 75 IU/L (28–100 IU/L), lipase 35 U/L (13–60 IU/L); and the total hCG level decreased to 762.0 mIU/mL. The patient’s symptoms continued to improve during her course of hospitalization. The patient was discharged with oral ciprofloxacin 500 mg per 12 h for discharge medication, on the 6th day of admission after significant clinical and laboratory improvement. The patient presented with complete resolution of symptoms and biochemical results during ambulatory follow-up. The follow-up transvaginal sonography revealed a decrease in the size of the hematoma and left adnexa mass (Fig. 4A, 4B).

Discussion
We report a rare case of MTX-induced pancreatitis in a patient with EP and treated with MTX. EP may be life threatening, and further treatment is determined based on the patient’s hemodynamic status, size of the ectopic mass, and level of beta-hCG. Medical treatment primarily uses the regimen of MTX [1]. Current regimens of MTX reduce the total MTX dose that patients receive compared with other indications such as chemotherapy [4]. Therefore, the incidence of therapy-related side effects is low, and most are minor side effects such as nausea, vomiting, stomatitis, and diarrhea [1]. Cases of MTX-induced pancreatitis are rare, and previous studies have reported a case of a 16-year-old girl with systemic lupus erythematosus acute necrotizing pancreatitis was attributed to a combination regimen involving MTX [12]. Another case report is of a 36-year-old woman who received MTX along with etoposide, actinomycin D, cyclophosphamide, and vincristine for suspected hydatidiform mole developed AP. Moreover, another case of a 10-year-old girl with acute lymphatic leukemia who was treated with vincristine, daunorubicin, L-asparaginase, intrathecal MTX, cyclophosphamide, and oral 6 MP and had MTX-induced pancreatitis has been reported [13]. However, AP after using MTX for the treatment of EP was not found in previously published studies and this report has very important implication that we should consider other differential diagnosis when we evaluate the patient suffering severe abdominal pain, treated with MTX due to EP. Cramp abdominal pain which is self-limiting and controlled with nonsteroidal anti-inflammatory agent is common side effect during MTX for an EP [15]. Also, If a patient with EP experiences severe abdominal pain after MTX therapy, clinicians would mostly suspect ectopic mass rupture, which will necessitate surgical treatment, and other differential diagnosis may be overlooked. However, if there is no evidence of an ectopic mass rupture, suspicion of other causes, laboratory tests, and imaging must be performed based on the clinical manifestations.
The side effects of MTX may be overlooked in the clinical settings of EP. If a patient with EP experiences abdominal pain after MTX therapy, clinicians would mostly suspect ectopic mass rupture, which will necessitate surgical treatment. However, if there is no evidence of an ectopic mass rupture, suspicion of other causes, laboratory tests, and imaging must be performed based on the clinical manifestations.
This case report has some limitations. For the diagnosis of an adverse drug reaction, the time relationship between the use of the drug and the occurrence of the reaction should be assessed. In addition, drug rechallenge should be considered [16]. However, since the patient received a single dose of MTX, there was no chance of rechallenge with drug.
Second, MTX has rarely been reported to be associated with the pancreatitis, and its possible etiology is unknown. Therefore, future studies and case reports are required in this area.
Although MTX-induced pancreatitis is a rare complication, it is important for clinicians to be aware of all possible complications of MTX because it is a widely used medical treatment for EP. We hope that this may aid in the evaluation of patients with severe abdominal pain after using MTX therapy.
