Abstract
-
Purpose
Endoscopic vacuum therapy (EVT) has emerged as a highly effective approach for managing gastrointestinal transmural defects and may offer advantages over traditional methods, such as stenting. This study evaluated the clinical outcomes of EVT for gastrointestinal transmural defects resulting from leakages, perforations, and fistulas.
-
Methods
We retrospectively reviewed patients who underwent EVT for gastrointestinal transmural defects at Ewha Womans University Medical Center between February 2018 and September 2025 and analyzed clinical outcomes, adverse events, and risk factors associated with adverse events.
-
Results
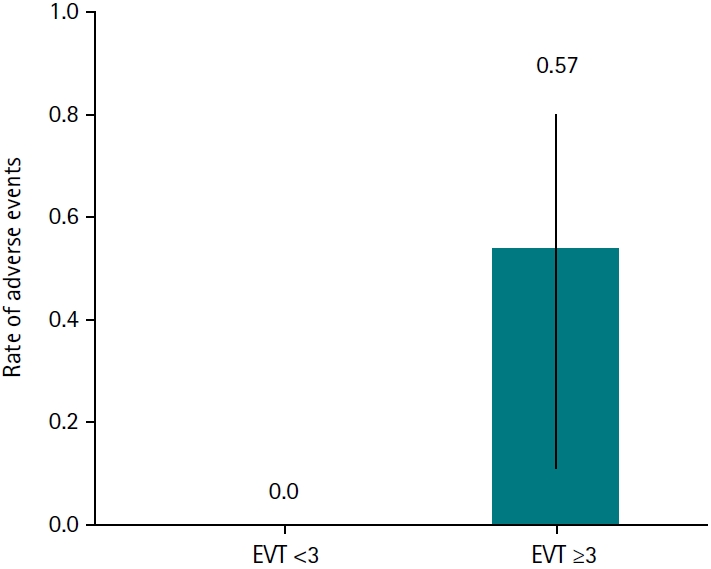
Fourteen patients were included (mean age, 63.9 years; 85.7% male). Stomach surgery was the most common etiology (50.0%), and malignancy accounted for 71.4% of cases. The median number of EVT sessions was 2.5, and the mean interval from the index event to the first EVT session was 10.5 days. EVT achieved a 100% technical success rate, with no 30-day mortality; there was 1 in-hospital death (7.1%), 2 cases of stricture (14.3%), and 1 major bleeding event (7.1%). Adverse events were observed more frequently in patients who underwent ≥3 EVT sessions (57.1%) compared with those who underwent <3 sessions, in whom no adverse events occurred.
-
Conclusion
This study suggests that EVT is a safe and effective treatment for gastrointestinal transmural defects, with high technical success rates. The number of EVT sessions and the timing of treatment initiation appeared to be associated with complications and overall clinical outcomes.
-
Keywords: Endoscopy; Gastrointestinal tract; Anastomotic leak; Therapeutics; Vacuum; Republic of Korea
Introduction
Endoscopic treatment of wall defects of the upper gastrointestinal (GI) tract, whether postoperative or iatrogenic, remains challenging for endoscopists [
1]. Several therapeutic approaches have been described, including endoscopic vacuum therapy (EVT), stenting, surgery, and conservative management [
2]. Among these options, EVT has been recognized as a highly effective method for managing leakage or perforation throughout the GI tract and represents one of the most important innovative techniques in the treatment of GI transmural defects [
3,
4].
EVT is an endoscopic approach that applies continuous negative pressure to the affected area, facilitating recovery by removing infectious secretions and promoting tissue regeneration [
5]. Compared with traditional stent placement therapy, EVT offers several advantages, including a reduced risk of infection and accelerated healing, while also minimizing stent-related complications such as migration and persistent leakage [
6]. In a recent network meta-analysis, EVT demonstrated significantly lower complication rates (odds ratio [OR], 0.23; 95% confidence interval [CI], 0.09–0.58) and mortality rates (OR, 0.43; 95% CI, 0.21–0.87) compared with stenting, whereas surgery was more frequently required for larger leaks than stenting (mean difference, 14.66; 95% CI, 4.61–24.70) [
2]. Other studies have reported favorable clinical outcomes, including a treatment success rate of 94.2% in a large-scale prospective study of 52 patients who underwent EVT for esophageal perforation or anastomotic leakage following esophagectomy or gastric cancer surgery [
7]. More recently, in Korea, the efficacy of EVT for anastomotic leaks following upper GI surgery has also been reported. In a cohort of 20 patients with anastomotic leakage after esophageal surgery, EVT achieved a treatment success rate of 95%, with a median fistula closure duration of 14.5 days. Preoperative chemotherapy was identified as a significant factor associated with a treatment duration exceeding 3 weeks [
8].
EVT has emerged as an effective treatment option for transmural gastrointestinal defects of various etiologies. However, data regarding risk factors for adverse events, such as stricture, bleeding, and infection, remain limited, and evidence supporting optimal treatment protocols is still lacking. Therefore, the primary objective of this study was to evaluate the success rate, clinical course, and adverse events associated with EVT. The secondary objectives were to identify risk factors associated with adverse events and to propose a safe and effective EVT protocol aimed at minimizing procedure-related risks.
Methods
Patient selection
This retrospective observational study was conducted at Ewha Womans University Medical Center. All patients who underwent EVT for leakage after GI surgery or foreign body impaction between February 2018 and September 2025 were included in this study. Exclusion criteria included cases in which EVT was performed for an aortoesophageal fistula after abdominal aortic aneurysm surgery, as well as cases in which initial vacuum therapy was performed intraoperatively for esophageal perforation.
The presence of leakage was determined based on computed tomography (CT) scanning and/or contrast swallowing examination and/or endoscopic identification of a defect. Medical records of eligible patients were reviewed, and clinical, radiological, and endoscopic data were collected. EVT was not applied to malignant tissue itself, and in patients with underlying malignancy, all procedures were restricted to leakage sites that were not directly associated with tumor involvement.
The study adhered to the principles outlined in the Declaration of Helsinki. The requirement for informed consent was waived following approval by the Institutional Review Board (IRB) of Ewha University Seoul Hospital (IRB approval no., 2024-10-062).
Definitions and outcome measures
The diagnosis of gastrointestinal transmural defects was based on clinical presentation and endoscopic and radiological findings, including esophagography, abdominal and pelvic CT, and chest CT. Clinical presentation was defined as physical findings such as abdominal pain, an increasing amount of drainage, fever, leukocytosis, and elevated C-reactive protein levels, or as changes in the content of the Hemovac drain. Endoscopic findings indicative of leakage consisted of direct visualization of a defect. Radiologic findings were defined as extraluminal contrast extravasation on esophagography, infiltration around the anastomotic site, and/or fistula tract formation on abdominal, pelvic, or chest CT.
Successful closure was defined as the absence of evidence of transmural defects on radiologic or endoscopic imaging after device removal, accompanied by the absence of clinical signs of persistent defects. Therapeutic outcomes of EVT were evaluated based on technical success, number of procedures, interval from defect diagnosis to EVT initiation, total indwelling duration, and indwelling period per exchange. Total indwelling duration was defined as the number of days from the first device insertion to final removal, whereas the indwelling period per exchange was defined as the number of days each EVT device remained in place before replacement. The interval between EVT procedures was defined as the number of days between consecutive EVT sessions, including from the first to the second session, the second to the third session, and subsequent sessions.
Major adverse events included procedure-related 30-day mortality, major bleeding requiring intervention or operative management, stricture formation within 1 year, and 1-year overall mortality. Procedure-related mortality was defined as death occurring before complete defect closure in the absence of other attributable causes.
Endoscopic vacuum therapy technique
EVT was performed under midazolam sedation. A single- or 2-channel gastroscope (GIF-HQ290, GIF-2T240, or GIF-2TQ260M; Olympus), a nasogastric tube, and a vacuum-assisted closure kit (CuraVAC; CGBio Inc.) were used. A polyurethane sponge was trimmed to match the size and location of the cavity, typically measuring ≤3.0×3.0 cm. The sponge was attached to the tip of the nasogastric tube and secured with silk sutures.
During the initial endoscopic examination, the defect and extraluminal cavity were irrigated. After identification of the leakage site, the sponge-mounted tube was advanced into the esophagus, and endoscopic guidance using grasping forceps was employed to ensure accurate placement of the sponge. The external end of the tube was subsequently connected to an electronic vacuum device, applying continuous negative pressure of −80 to −100 mm Hg. The final tube position was marked at the nostrils and secured to prevent displacement.
The sponge was exchanged every 1 to 2 days, depending on the patient’s clinical condition. If endoscopic examination suggested complete closure of the defect, the sponge was removed, and esophagography or esophagogastroduodenoscopy (EGD) was performed to confirm healing.
Although the fundamental principles of EVT were applied consistently, including device selection, continuous negative pressure application, and regular sponge exchange, key procedural parameters such as negative pressure intensity, sponge exchange intervals, and total treatment duration were adjusted according to individual patient conditions, defect characteristics, and clinical responses.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics ver. 26.0 (IBM Corp.). Continuous variables are presented as mean±standard deviation (SD) or median (range), and categorical variables as counts and percentages. The association between the number of EVT sessions and complications was evaluated by stratifying patients into 2 groups, with comparisons performed using Fisher’s exact test (2-sided). Statistical significance was defined as P<0.05.
Results
Clinical characteristics and endoscopic vacuum therapy–related parameters
Fourteen patients were included in the study. The mean age was 63.9±6.8 years, and 12 patients (85.7%) were male. The most common etiology of perforation was stomach surgery (n=7; 50.0%), followed by esophageal surgery (n=5; 35.7%) and esophageal foreign body impaction (n=2; 14.3%). Regarding underlying disease, 10 patients (71.4%) had malignant conditions and 4 patients (28.6%) had benign conditions. Leakage was most frequently diagnosed by esophagography (n=6; 42.9%), followed by EGD (n=3; 21.4%), chest CT (n=3; 21.4%), and abdominal CT (n=2; 14.3%). The overall treatment success rate was 100% (14/14), with all patients achieving defect closure without requiring surgical revision or an alternative intervention. The median number of EVT sessions was 2.5 (range, 1–6), and the mean interval from surgery to the first EVT session was 10.5±8.7 days. The mean duration of EVT indwelling was 7.0±2.5 days (range, 2–15 days) (
Table 1).
There was no 30-day mortality (0%). One patient (7.1%) experienced in-hospital mortality due to sepsis secondary to pneumonia, which was considered unrelated to the EVT procedure or gastrointestinal leakage. Adverse events occurred in a subset of patients: stricture developed in 2 patients (14.3%), and major bleeding occurred in 1 patient (7.1%) (
Table 2).
Among the 14 patients, the incidence of adverse events differed according to the number of EVT sessions performed. No adverse events occurred in patients who underwent fewer than 3 EVT sessions (0/7; 0.0%), whereas 4 of 7 patients (57.1%) who underwent 3 or more EVT sessions developed adverse events (
Fig. 1). The rate of adverse events was higher in the ≥3 EVT session group (risk ratio, 8.57; 95% CI, 0.55–134.6). Adverse events were more frequently observed in patients who underwent ≥3 EVT sessions than in those who underwent <3 sessions (57.1% vs. 0%), demonstrating a trend toward statistical significance (2-sided P=0.07).
Discussion
This retrospective study demonstrated that EVT achieved complete defect closure in all 14 patients, with no 30-day mortality. Adverse events consisted of 2 cases of stricture and 1 case of major bleeding. Notably, the risk of adverse events increased with the number of EVT sessions, as patients who underwent 3 or more sessions exhibited a substantially higher rate of adverse events. These findings support the efficacy and safety of EVT, while suggesting that patients requiring multiple sessions should be regarded as a higher-risk subgroup.
Our findings are consistent with those of previous studies reporting high technical success rates and acceptable safety profiles of EVT in the treatment of gastrointestinal transmural defects [
9,
10]. Most prior reports have focused primarily on overall efficacy, whereas risk factors for adverse events have been less systematically examined. In this context, the present study provides additional insight by identifying the number of EVT sessions as a potential predictor of adverse events. Patients requiring repeated EVT sessions may have larger or more complex defects, delayed tissue healing, or unfavorable clinical conditions, all of which may increase susceptibility to complications.
In the present cohort, 1 patient experienced major bleeding following EVT, necessitating subsequent embolization. Although such events are uncommon, uncontrollable hemorrhage represents a major complication associated with EVT [
7,
11]. The risk of bleeding may be particularly elevated in cases involving tracheoesophageal fistula, in which proximity to major vascular structures increases the potential for catastrophic outcomes [
12,
13]. To mitigate this risk, preprocedural evaluation of adjacent vessels using contrast-enhanced CT may be considered to identify vulnerable anatomy [
14]. In selected high-risk situations, avoiding continuous negative pressure and allowing passive drainage may serve as precautionary strategies. These considerations underscore the importance of individualized risk assessment and tailored EVT application in clinical practice.
The timing of EVT initiation has been proposed as a factor that may influence treatment outcomes. However, the present study was limited in its ability to define ‘early initiation’ or to establish an optimal timing threshold. Previous studies have reported high success rates and relatively short EVT indwelling durations in cases of acute iatrogenic perforation, which may be attributable to prompt defect recognition and treatment initiation within 24 hours [
15,
16]. Owing to the limited number of cases, subgroup analyses and statistically robust conclusions could not be derived in the present study. Nevertheless, we observed a trend suggesting that longer intervals between the defect event and the first EVT session were associated with a higher risk of adverse events. These findings should be interpreted as exploratory observations that are broadly consistent with existing literature.
EVT has demonstrated high clinical and technical success rates in the management of GI transmural defects, with the added advantage of enabling simultaneous drainage of intraluminal and extraluminal collections. Nevertheless, patient discomfort related to the transnasal drainage tube and the need for repeated endoscopic procedures remain notable limitations [
1,
17,
18]. Accordingly, increasing attention has been directed toward preventive strategies aimed at reducing postoperative defects following upper GI surgery. One such approach is preemptive EVT, which involves prophylactic application of EVT before defect formation to reduce the risk of adverse events and improve outcomes [
19,
20]. Recent studies have highlighted preemptive EVT as a promising strategy with potential for broader clinical adoption. Our findings suggest that earlier initiation of EVT may be associated with more favorable outcomes, although confirmation in well-designed prospective studies is required.
This study has several limitations. First, this was a single-center case series involving only 14 patients, which limits the generalizability of the findings. The small sample size may have resulted in insufficient statistical power to detect subtle between-group differences, underscoring the need for validation in larger, multicenter prospective studies. Second, the retrospective design introduces inherent risks of selection bias, incomplete data capture, and unmeasured confounding. Third, the absence of direct comparisons with alternative treatment modalities, such as stent placement or surgical revision, precludes definitive conclusions regarding the relative efficacy of EVT. Fourth, standardized data regarding defect size and anatomical location were not consistently available, limiting assessment of the independent effects of defect-related factors on outcomes and adverse events.
In conclusion, this study suggests that EVT is an effective and safe treatment modality for gastrointestinal transmural defects, achieving complete closure in all cases with low short-term mortality. The number of EVT sessions was identified as a factor associated with complication occurrence, and a trend toward more favorable outcomes was observed with earlier EVT initiation. However, given the limited sample size, these findings should be interpreted as exploratory. Preemptive EVT may represent a promising strategy for reducing postoperative complications; however, its clinical utility and optimal indications require validation in rigorously designed prospective studies.
-
Authors’ contribution
Concept and design: KNS, ARC. Data analysis and interpretation: KNS, ARC, CHT, JRB. Drafting of the manuscript: ARC. Critical revision of the manuscript for essential intellectual content: KNS, ARC, CHT, JRB, EMS, SAJ. All authors have read and approved the final manuscript.
-
Conflict of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
Data cannot be shared due to issues related to the identification of vulnerable participants.
-
Acknowledgments
None.
-
Supplementary materials
None.
Fig. 1.Rate of adverse events according to the number of endoscopic vacuum therapy (EVT) sessions. Adverse events were more frequently observed in patients who underwent ≥3 EVT sessions than in those who underwent <3 sessions (57.1% vs. 0%), demonstrating a trend toward statistical significance (2-sided P=0.07). Error bars represent 95% confidence intervals for adverse event rates.
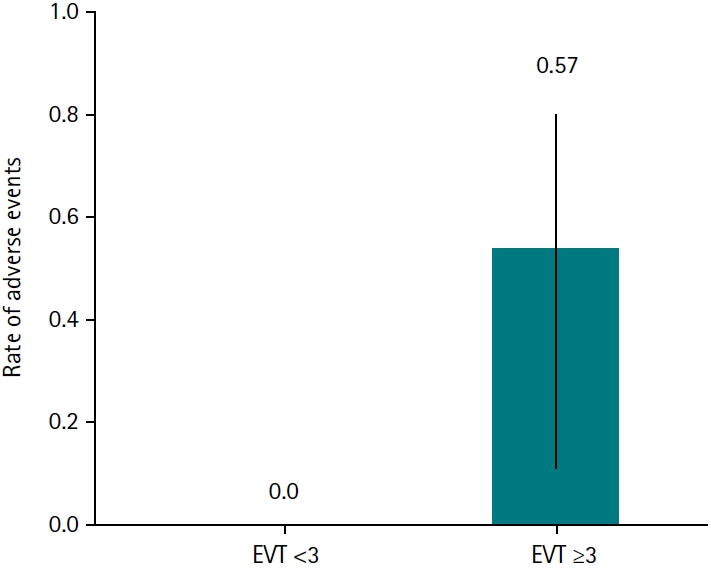
Table 1.Clinical characteristics and endoscopic vacuum therapy–related parameters
|
Characteristic |
Value |
|
Age (yr) |
63.9±6.8 |
|
Sex, male |
12 (85.7) |
|
Etiology of defects |
|
|
Stomach surgery |
7 (50.0) |
|
Esophageal surgery |
5 (35.7) |
|
Foreign body |
2 (14.3) |
|
Cause |
|
|
Malignancy |
10 (71.4) |
|
Benign |
4 (28.6) |
|
Defects diagnosis method |
|
|
Esophagography |
6 (42.9) |
|
EGD |
3 (21.4) |
|
Chest CT |
3 (21.4) |
|
Abdominal CT |
2 (14.3) |
|
EVT treatment |
|
|
EVT success |
14 (100.0) |
|
Sponge exchange |
2.5 (1–6) |
|
Time to first EVT from defect event (day) |
10.5±8.7 (0–35) |
|
Interval between EVT procedures |
7.0±2.5 (2–15) |
|
Total EVT indwelling duration (day) |
22.1±14.5 (6–50) |
Table 2.Adverse events after endoscopic vacuum therapy
|
Adverse events |
No. (%) |
|
30-day mortality |
0 (0.0) |
|
In-hospital mortality |
1 (7.1) |
|
Stricture |
2 (14.3) |
|
Massive bleeding |
1 (7.1) |
References
- 1. Kouladouros K. Applications of endoscopic vacuum therapy in the upper gastrointestinal tract. World J Gastrointest Endosc 2023;15:420-433. https://doi.org/10.4253/wjge.v15.i6.420
- 2. Murray W, Davey MG, Robb W, Donlon NE. Management of esophageal anastomotic leaks, a systematic review and network meta-analysis. Dis Esophagus 2024;37:doae019. https://doi.org/10.1093/dote/doae019
- 3. Loske G, Schorsch T, Muller C. Endoscopic vacuum sponge therapy for esophageal defects. Surg Endosc 2010;24:2531-2535. https://doi.org/10.1007/s00464-010-0998-x
- 4. Weidenhagen R, Gruetzner KU, Wiecken T, Spelsberg F, Jauch KW. Endoscopic vacuum-assisted closure of anastomotic leakage following anterior resection of the rectum: a new method. Surg Endosc 2008;22:1818-1825. https://doi.org/10.1007/s00464-007-9706-x
- 5. Le TM, Tran VH, Chung KS, Jeon SW. Endoscopic vacuum therapy for gastrointestinal transmural defects: a literature review. Clin Endosc 2025;58:181-190. https://doi.org/10.5946/ce.2024.150
- 6. Vohra I, Gopakumar H, Sharma NR, Puli SR. Efficacy of endoscopic vacuum therapy in esophageal luminal defects: a systematic review and meta-analysis. Clin Endosc 2025;58:53-62. https://doi.org/10.5946/ce.2023.282
- 7. Laukoetter MG, Mennigen R, Neumann PA, Dhayat S, Horst G, Palmes D, Senninger N, Vowinkel T. Successful closure of defects in the upper gastrointestinal tract by endoscopic vacuum therapy (EVT): a prospective cohort study. Surg Endosc 2017;31:2687-2696. https://doi.org/10.1007/s00464-016-5265-3
- 8. Min YW, Kim T, Lee H, Min BH, Kim HK, Choi YS, Lee JH, Rhee PL, Kim JJ, Zo JI, Shim YM. Endoscopic vacuum therapy for postoperative esophageal leak. BMC Surg 2019;19:37. https://doi.org/10.1186/s12893-019-0497-5
- 9. Jung DH, Yun HR, Lee SJ, Kim NW, Huh CW. Endoscopic vacuum therapy in patients with transmural defects of the upper gastrointestinal tract: a systematic review with meta-analysis. J Clin Med 2021;10:2346. https://doi.org/10.3390/jcm10112346
- 10. Intriago JM, de Moura DT, do Monte Junior ES, Proença IM, Ribeiro IB, Sanchez-Luna SA, Bernardo WM, de Moura EG. Endoscopic vacuum therapy (EVT) for the treatment of post-bariatric surgery leaks and fistulas: a systematic review and meta-analysis. Obes Surg 2022;32:3435-3451. https://doi.org/10.1007/s11695-022-06228-0
- 11. Pournaras DJ, Hardwick RH, Safranek PM, Sujendran V, Bennett J, Macaulay GD, Hindmarsh A. Endoluminal vacuum therapy (E-Vac): a treatment option in oesophagogastric surgery. World J Surg 2018;42:2507-2511. https://doi.org/10.1007/s00268-018-4463-7
- 12. Scognamiglio P, Reeh M, Melling N, Kantowski M, Eichelmann AK, Chon SH, El-Sourani N, Schon G, Holler A, Izbicki JR, Tachezy M. Management of intra-thoracic anastomotic leakages after esophagectomy: updated systematic review and meta-analysis of endoscopic vacuum therapy versus stenting. BMC Surg 2022;22:309. https://doi.org/10.1186/s12893-022-01764-z
- 13. de Moura DT, Hirsch BS, Ribas PH, Silveira SQ, Guedes HG, Bestetti AM. Endoscopic vacuum therapy: pitfalls, tips and tricks, insights, and perspectives. Transl Gastroenterol Hepatol 2024;9:50. https://doi.org/10.21037/tgh-23-86
- 14. Little BP, Mendoza DP, Fox A, Wu CC, Ackman JB, Shepard JA, Muniappan A, Digumarthy SR. Direct and indirect CT imaging features of esophago-airway fistula in adults. J Thorac Dis 2020;12:3157-3166. https://doi.org/10.21037/jtd-20-244
- 15. Sendino O, Loras C, Mata A, Momblan D, Andujar X, Cruz M, Cardenas A, Marquez I, Uchima H, Cordova H, de Lacy AM, Espinos J. Safety and efficacy of endoscopic vacuum therapy for the treatment of perforations and anastomotic leaks of the upper gastrointestinal tract. Gastroenterol Hepatol 2020;43:431-438. https://doi.org/10.1016/j.gastrohep.2020.01.019
- 16. Singh RR, Nussbaum JS, Kumta NA. Endoscopic management of perforations, leaks and fistulas. Transl Gastroenterol Hepatol 2018;3:85. https://doi.org/10.21037/tgh.2018.10.09
- 17. Medas R, Rodrigues-Pinto E. Technical review on endoscopic treatment devices for management of upper gastrointestinal postsurgical leaks. Gastroenterol Res Pract 2023;2023:9712555. https://doi.org/10.1155/2023/9712555
- 18. Livingstone I, Pollock L, Sgromo B, Mastoridis S. Current status of endoscopic vacuum therapy in the management of esophageal perforations and post-operative leaks. Clin Endosc 2021;54:787-797. https://doi.org/10.5946/ce.2021.240
- 19. Muller PC, Vetter D, Kapp JR, Gubler C, Morell B, Raptis DA, Gutschow CA. Pre-emptive endoluminal negative pressure therapy at the anastomotic site in minimally invasive transthoracic esophagectomy (the preSPONGE Trial): study protocol for a multicenter randomized controlled trial. Int J Surg Protoc 2021;25:7-15. https://doi.org/10.29337/ijsp.24
- 20. Adamenko O, Ferrari C, Seewald S, Schmidt J. Prophylactic endoluminal vacuum therapy after major gastrointestinal surgery: a systematic review. Updates Surg 2022;74:1177-1186. https://doi.org/10.1007/s13304-022-01265-x