Abstract
-
Purpose
Human papillomavirus is the dominant etiological factor underlying atypical cervical squamous epithelial cell abnormalities and cervical carcinoma. Currently, only a limited number of drugs targeting specific biomarkers in cervical cancer are available. This study aimed to assess the expression of estrogen receptor (ER), human epidermal growth factor receptor 2 (HER2), and the Ki-67 proliferative index (Ki-67) in p16-positive cervical squamous premalignant and malignant lesions, which may help clarify the potential role of targeted therapies in cervical cancer.
-
Methods
In p16-positive, histologically proven premalignant and malignant cervical lesions, ER, HER2, and Ki-67 expression were evaluated according to predefined criteria.
-
Results
p16 showed strong nuclear and cytoplasmic positivity in 54 of 56 cases. Patchy nuclear positivity was mainly observed in low-grade squamous intraepithelial lesion (LSIL) cases (2/56). Ki-67 demonstrated a variable proliferative index ranging from 5% to 95% across all cases, with higher indices predominantly observed in squamous cell carcinomas (SCC). ER positivity in LSIL, high-grade squamous intraepithelial lesion, and SCC was 100% (2/2), 66.7% (10/15), and 46.15% (18/39), respectively. HER2 expression was predominantly negative, observed in 78.6% (44/56) of cases, equivocal in 17.8% (10/56), and positive in 3.6% (2/56). Both HER2-positive cases were SCC. ER and HER2 interpretations were analyzed and were not significantly correlated with clinical or pathological parameters.
-
Conclusion
ER positivity decreased with progression of cervical squamous lesions, and HER2 expression was rare in cervical squamous neoplasia. No statistically significant correlation was identified between ER or HER2 expression and clinicopathological parameters. The findings of the current study may help fill gaps in the existing literature and provide essential foundational knowledge for optimizing emerging therapeutic strategies, including ER- and HER2-related therapies.
-
Keywords: Cervix; Estrogen receptors; HER2; p16; Squamous intraepithelial lesions; Uterine cervical neoplasms
Introduction
Worldwide, cervical cancer is the fourth most common cancer and the fourth leading cause of cancer-related death among women, as reported by GLOBOCAN (Global Cancer Observatory) [
1]. Human papillomavirus (HPV) infection is a requisite etiological factor for HPV-associated cervical cancer, although it is not sufficient on its own. Other important cofactors, including certain sexually transmitted diseases, smoking, increased parity, and prolonged oral contraceptive use, are also associated with the development of cervical cancer [
1].
The World Health Organization categorizes cervical squamous cell carcinomas (SCC) into HPV-associated and HPV-independent types. However, the vast majority of cervical SCCs are HPV-associated, with only rare cases reported as HPV-independent [
2]. Overexpression of p16 serves as a reliable surrogate biomarker for HPV infection and can be detected using immunohistochemistry [
2]. Ki-67 is a marker of cellular proliferation, and increased Ki-67 expression is associated with aggressive tumor behavior and poorer clinical outcomes, making it a useful prognostic biomarker [
3]. Estrogen facilitates the proliferation and differentiation of cervical epithelial cells through estrogen receptor alpha (ERα), as estrogen levels rise during the menstrual cycle, leading to hyperplastic epithelium without pathological alteration [
4]. A mouse-based study demonstrated the essential role of ERα in mediating the carcinogenic effects of estrogen in the cervix. ERα-sufficient HPV transgenic mice developed progressive atypical squamous metaplasia that culminated in cervical cancer following estrogen treatment [
4]. Furthermore, ERα antagonist drugs showed significant efficacy in eradicating pre-existing cervical cancers, suggesting the potential of these agents in preventing cervical cancer development [
4]. Human epidermal growth factor receptor 2 (HER2) is a membrane-bound receptor with intrinsic tyrosine kinase activity and plays a key role in cellular transformation, carcinogenesis, and maintenance of malignant phenotypes [
5,
6]. These effects occur through activating mutations, amplification of the
HER2 gene, or overexpression of the HER2 protein, making HER2 a prime target for antitumor therapies [
5,
6]. The role of HER2 expression in gynecological malignancies remains under investigation, and consensus has not yet been established [
5,
6]. However, both preclinical models and clinical studies suggest that somatic
HER2 gene mutations in cervical cancer represent promising targets for selective inhibitors [
5,
6]. Uterine cervical carcinomas have a paucity of clinically effective targetable biomarkers [
7]. Estrogen receptor (ER), HER2, and fibroblast activation protein have emerged as potential therapeutic or theranostic targets across several gynecologic and genitourinary malignancies [
7]. Currently, only a limited number of cervical cancer-specific targetable biomarkers are available, such as pembrolizumab therapy guided by programmed cell death ligand 1 (PD-L1) expression [
7].
While p16 and Ki-67 are well-established biomarkers in cervical lesions, the expression patterns and clinical significance of ER and HER2 in p16-positive cervical squamous lesions remain poorly characterized. Identification of these potentially targetable biomarkers may open new avenues for precision medicine in cervical cancer. In this study, we aimed to evaluate the expression of ER and HER2 as potential therapeutic targets, along with Ki-67 as a proliferation marker, in p16-positive cervical squamous premalignant and malignant lesions, and to assess their association with clinicopathological parameters.
Methods
Study design and setting
The present study was a cross-sectional, hospital-based study conducted over a period of 2 years in the Department of Pathology and Laboratory Medicine, in collaboration with the Department of Obstetrics and Gynecology, at All India Institute of Medical Science, Bibinagar, India. The sample size was calculated using the formula. S=Z2pqL2, where S represents the sample size, Z is the Z score, p is the population proportion, q is (1−p), and L is the relative precision. Based on this calculation, the final sample size was 56.
After the study was approved by the Institutional Ethics Committee (IEC Ref No: AIIMS/BBN/IEC/JUNE/2023/284), detailed clinical data were collected, including age, age at menarche, last menstrual period, educational status, parity, age at first delivery, and presenting symptoms such as white discharge, vaginal bleeding, foul-smelling discharge, pain, history of weight loss, and clinical diagnosis.
Initial cohort
The study population comprised all patients with suspected cervical lesions who underwent biopsy or hysterectomy according to the Department of Obstetrics and Gynecology treatment protocol during the study period and were reported as having premalignant or malignant ectocervical squamous lesions. Cervical biopsy and hysterectomy specimens were received in formalin in the Department of Pathology for histopathological examination. The cases were evaluated and categorized as premalignant low-grade squamous intraepithelial lesion (LSIL)/cervical intraepithelial neoplasia (CIN) 1, high-grade squamous intraepithelial lesion (HSIL)/CIN 2 or CIN 3, or malignant lesions. Wherever applicable, pathological tumor–node–metastasis staging was performed.
In the study population of premalignant and malignant ectocervical squamous lesions, all cases were subjected to ready-to-use p16 immunohistochemical staining using a mouse monoclonal antibody (Clone JC8). For positive p16 staining, the localization pattern was assessed as nuclear, cytoplasmic, or nucleocytoplasmic. The distribution of staining was analyzed based on intensity, categorized as weak (1+), moderate (2+), or strong (3+). Positivity in 5%–25% of tumor cells was considered grade 1, 26%–50% grade 2, and ≥51% grade 3.
Exclusion criteria
Specimens that were inadequate for processing, patients who had already received or were undergoing treatment, non-squamous atypical cervical lesions, and patients receiving chemotherapy or radiotherapy for any other malignancy were excluded from the study.
Final analyzed cases
All p16-positive premalignant and malignant squamous cell lesions of the cervix were further evaluated for ER, HER2, and Ki-67 immunoexpression using ready-to-use immunohistochemical markers for Ki-67 (Clone MIB-1, mouse monoclonal antibody), ER (Clone EP1, rabbit monoclonal antibody), and HER2 (Clone EP3, rabbit monoclonal antibody). Interpretation of ER and HER2 staining in cervical carcinoma followed the standard protocol used for hormone receptor assessment in breast carcinoma.
For ER analysis in CIN lesions, expression was recorded based on localization within the epithelium, specifically the lower one-third, middle two-thirds, or full thickness. Both staining intensity and the percentage of positive tumor cells were documented, and an Allred score was calculated. Scores of 0–2 were considered ER-negative, whereas scores >3 were considered ER-positive. The same Allred scoring system was applied to SCC cases.
For HER2 analysis, a score of 0 was assigned for absent staining or weak staining in <10% of tumor cells, score 1 for incomplete weak membranous staining in ≥10% of tumor cells, score 2 for complete weak to moderate membranous staining, and score 3 for complete strong membranous staining in >10% of tumor cells. Scores of 0 and 1 were considered negative, score 2 equivocal, and score 3 positive.
Ki-67, a nuclear proliferation marker, was assessed in the area of maximum expression and calculated as the percentage of atypical squamous cells demonstrating positive nuclear staining. In CIN lesions, Ki-67 expression was categorized according to involvement of the epithelium’s lower one-third, middle two-thirds, or full thickness. In SCC cases, the percentage positivity was recorded and dichotomized as ≤60% or >60%, based on the approximate mean Ki-67 labeling index, for statistical analysis.
Statistical analysis
Data were analyzed using IBM SPSS for Windows ver. 28.0 (IBM Corp.). Categorical variables are presented as frequencies and percentages. Associations between categorical variables (ER/HER2 status versus clinicopathological parameters and p16/Ki-67 expression) were assessed using the Pearson chi-square test. All subgroup analyses, particularly those involving small sample sizes (e.g., LSIL, n=2), were exploratory and hypothesis-generating; therefore, nonsignificant findings were interpreted with caution in light of the study’s limitations.
Results
Out of 64 cases comprising premalignant and malignant ectocervical lesions evaluated during the study period, 8 cases were p16-negative and were excluded from further analysis. The remaining 56 samples showed block positivity for p16, and in these cases, the expression patterns of ER, HER2, and Ki-67 immunomarkers, along with their associations with clinicopathological parameters, were assessed.
In the study population, age ranged from 25 to 83 years, with a mean age of 51.39 years. The mean age for LSIL, HSIL, and SCC progressively increased with lesion severity and was 36.5, 42.2, and 55.69 years, respectively. Overall, 71.4% of women were older than 45 years, indicating that cervical lesions were more commonly diagnosed in middle-aged and older women.
Early menarche (≤12 years) was observed in 62.5% of cases, and early childbearing (≤18 years) was noted in 50% of women. At presentation, 60.7% of women were postmenopausal. A high illiteracy rate of 67.9% (38/56) was observed in the study cohort. The most common presenting symptom was vaginal bleeding (96.4%), followed by white discharge and pain. Women with early menarche, early childbearing, lack of formal education, age >45 years, and postmenopausal status showed a higher frequency of SCC (
Table 1).
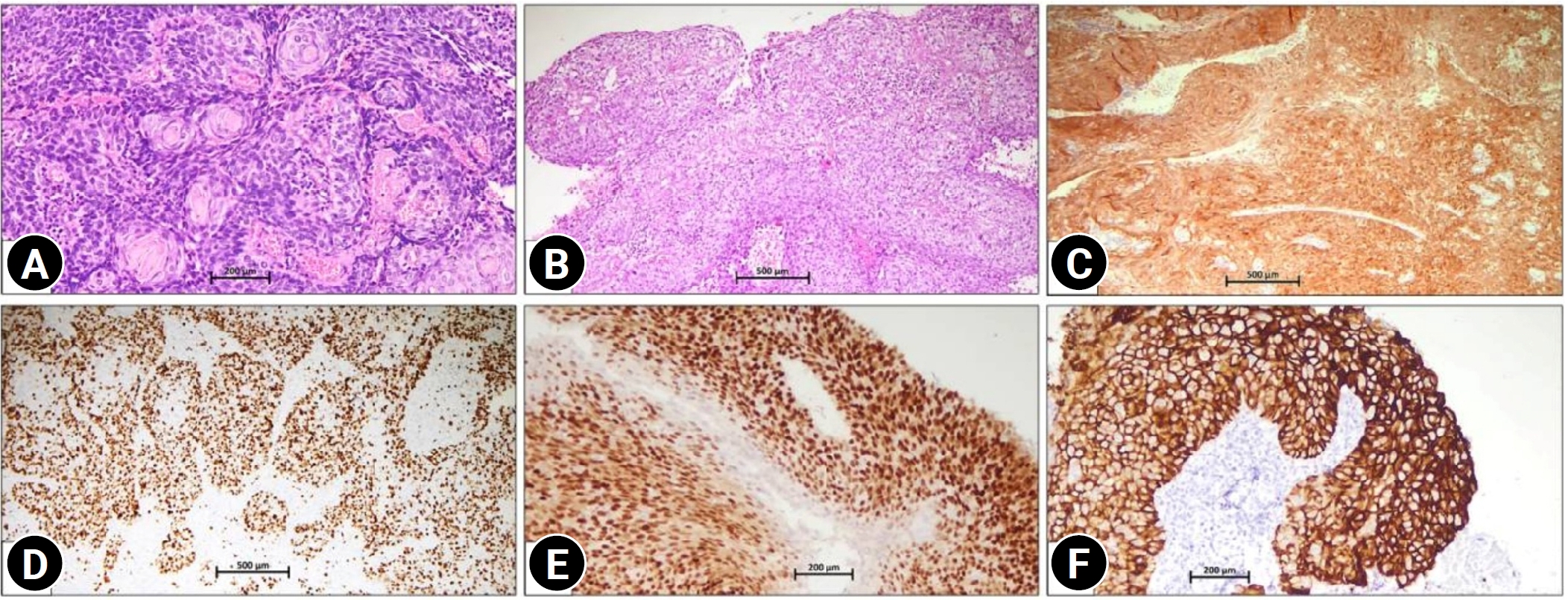
The most common histological diagnosis was SCC (39/56), followed by HSIL (15/56). Among the 39 SCC cases, the non-keratinizing subtype was most frequent, accounting for 66.7%, including 3 well-differentiated, 22 moderately differentiated, and 1 poorly differentiated tumors (
Fig. 1A,
B). Among keratinizing SCC, 1 case was well-differentiated and 12 were moderately differentiated. Seven hysterectomy specimens from cases initially diagnosed as SCC on cervical biopsy were received. The greatest tumor dimension averaged 1.75±1.28 cm, with a range of 0.5–4 cm. Stromal invasion measured 1 mm and 3 mm in 1 patient each and exceeded 5 mm in 5 patients. Lymphovascular invasion and parametrial involvement were each observed in 1 patient. Pathological staging showed pT1b1 in 4 patients, pT1a1 in 2 patients, and pT2b in 1 patient. No regional lymph node or distant metastasis was identified, or such information was unavailable.
p16 demonstrated strong expression across the cohort, supporting HPV-related pathogenesis. Nuclear-cytoplasmic block-type positivity was observed in 96.4% (54/56) of cases. Patchy nuclear positivity was mainly identified in LSIL lesions (2/56) (
Fig. 1C). The intensity and grade of p16 expression showed an increasing trend with advancing lesion severity (
Table 1).
The proliferation marker Ki-67 showed a variable proliferative index ranging from 5% to 95% (
Fig. 1D). Among the 17 patients with CIN, Ki-67 expression was limited to the lower one-third of the epithelium in 5 cases, extended to the middle two-thirds in 8 cases, and involved the full thickness in 4 cases. Among the 39 SCC cases, 53.8% (21/39) demonstrated a high proliferative index (>60%), whereas 46.2% (18/39) showed a proliferative index ≤60% (
Table 1).
ER (
Fig. 1E) positivity was noted in 53.6% (30/56) of cervical lesions, with expression levels ranging from 0% to >66%.
Regarding intensity, strong expression was observed in 17.9% (10/56) of cases, while 25.0% (14/56) demonstrated moderate intensity (
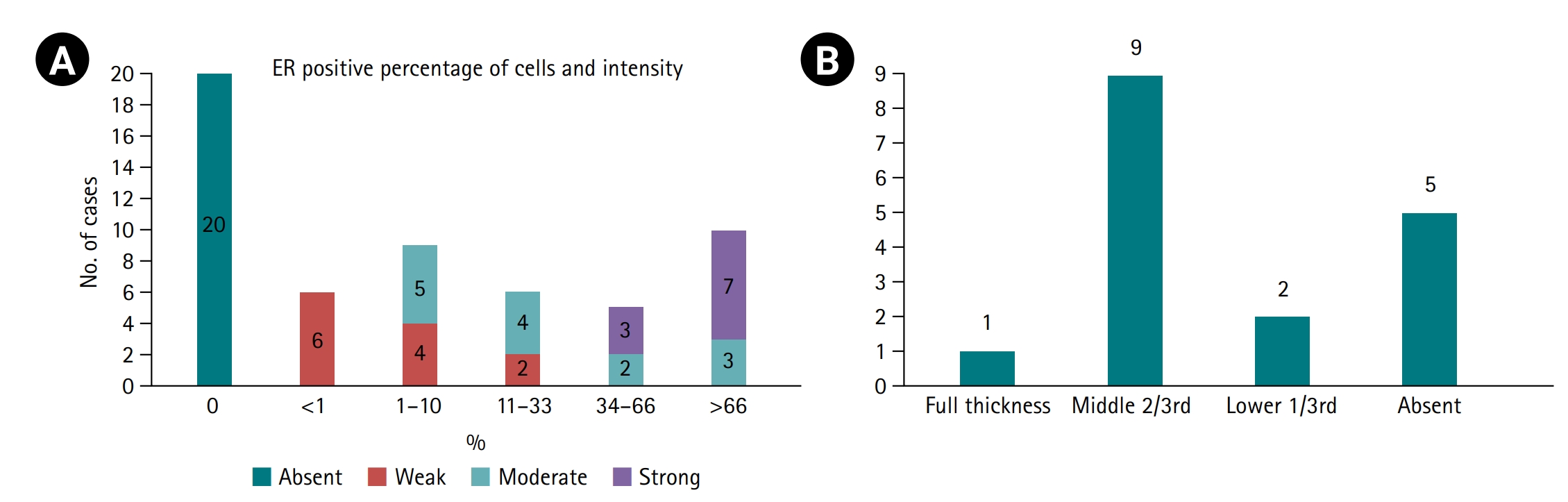
Table 1). In CIN cases, 52.9% (9/17) showed ER expression extending up to the middle two-thirds of the epithelium (
Fig. 2). A diffuse expression pattern was observed in 47.1% (8/17) of CIN cases, whereas a focal pattern was noted in 23.5% (4/17), and ER expression was absent in 29.4% (5/17) (
Fig. 2). Overall, ER positivity in LSIL, HSIL, and SCC was 100%, 66.7%, and 46.15%, respectively.
HER2 expression was predominantly negative in 78.6% (44/56) of cases, equivocal in 17.8% (10/56), and positive in 3.6% (2/56) (
Fig. 1F). Both HER2-positive cases were SCC and were associated with early menarche; one tumor was keratinizing and the other non-keratinizing. HER2 expression was negative in all well-differentiated and poorly differentiated tumors. Among moderately differentiated tumors, 2 cases were HER2 positive, 7 were equivocal, and 25 were negative.
ER and HER2 expression statuses were analyzed in relation to clinicopathological parameters, including age at presentation, age at menarche, menopausal status, parity, age at first delivery, clinical presentation, histological diagnosis, and degree of p16 and Ki-67 expression. The analysis demonstrated that neither ER nor HER2 status showed a statistically significant correlation with the evaluated clinical or pathological parameters. Additionally, no significant correlation was identified between ER and HER2 expression (
Table 2).
Discussion
The present study included 56 cases of p16-positive cervical lesions, with an age range of 25 to 83 years. Previous studies have reported the mean age for cervical lesions to range from 34.59 to 52.28 years [
8-
13]. In the present study, the overall mean age was 51.39 years, and the mean age for LSIL, HSIL, and SCC progressively increased with lesion severity, measuring 36.5, 42.2, and 55.69 years, respectively. Consistent with prior observations, most LSIL cases occurred before the age of 45 years, whereas the majority of HSIL and SCC cases occurred after the age of 45 years [
8,
10,
12,
14]. The observed variation in age distribution may be attributable to the relatively small sample size or differences in patient selection.
Bhadauria et al. [
15] reported a mean age at childbearing of 18.9 years among SCC cases, whereas in the present study, the mean age at childbearing was 17.9 years across all cases. All SCC cases in the present study occurred in multiparous women and predominantly among those who attained menarche at ≤12 years or had an age at first delivery of ≤18 years. Early menarche and early age at first delivery may reflect prolonged lifetime estrogen exposure and early onset of sexual activity, respectively, while multiparity may further contribute as a risk factor for cervical dysplasia.
Carcinomas are reported more frequently in postmenopausal women, whereas precursor lesions are more common in premenopausal women [
8,
10,
13,
16,
17]. In the present study, SCC predominated among postmenopausal women, while LSIL and HSIL were more frequently observed in premenopausal women. The high illiteracy rate observed in this study was comparable to that reported by Bhadauria et al. [
15], which may indicate limited awareness of cancer symptoms and screening practices among illiterate populations. The most common presenting symptom was bleeding per vaginam in SCC and HSIL cases, whereas white discharge was most frequently reported in LSIL cases, findings that are consistent with those reported by Singh et al. [
8] and Mehdi et al. [
16]. These symptom patterns may reflect increased vascularity and epithelial disruption in dysplastic and malignant lesions.
The reported proportions of LSIL, HSIL, and SCC among cervical lesions vary widely in the literature, ranging from 4.8% to 35.7%, 20.2% to 86.5%, and 17.2% to 77.4%, respectively [
5,
8,
9,
13,
14,
16,
18-
27]. Similarly, the reported frequencies of well-differentiated, moderately differentiated, and poorly differentiated SCC range from 16% to 67.7%, 17.4% to 64%, and 9.8% to 33.2%, respectively [
10,
13,
15-
17,
27,
28]. Variability has also been reported with respect to histological subtype, including keratinizing and non-keratinizing SCC [
13,
15,
17,
28]. The very low number of LSIL cases and the predominance of moderately differentiated and non-keratinizing SCC observed in the present study may be attributable to the limited sample size or demographic variation within the study population.
The intensity and grading patterns of p16 staining have been variably reported in the literature, and the findings of the present study fall within these reported ranges [
8,
9,
11,
13,
18,
19]. HSIL and SCC predominantly showed nuclear-cytoplasmic, block-type positivity, whereas LSIL demonstrated patchy nuclear positivity, findings that are consistent with previous studies [
8,
16]. p16 is a tumor suppressor protein that is overexpressed in HPV-related lesions, while CIN 1 often shows variable expression with a substantial proportion of negative cases. Previous studies have reported p16-negative expression in 0%–84% of CIN 1/LSIL, 1.1%–50% of CIN 2, 0.6%–14% of CIN 3, and 0%–20% of SCC cases [
8,
9,
13]. Grade 1 p16 expression has been reported in 10%–20.6% of CIN 1/LSIL, 6.3%–20% of CIN 2, 2.5%–10% of CIN 3, and 0%–18% of SCC cases [
8,
9,
13]. In the present study, grade 1 expression was observed in 100% of LSIL cases and 6.7% of HSIL cases. Grade 2 expression has been reported in 0%–21% of CIN 1, 4.5%–24% of CIN 2, 1.2%–40% of CIN 3, and 0%–24% of SCC cases [
8,
9,
13]; in the present study, 2.6% of SCC cases and 26.7% of HSIL cases showed grade 2 expression. Grade 3 expression has been reported in 0%–70% of CIN 1/LSIL, 6%–86.9% of CIN 2, 36%–95.7% of CIN 3, 100% of HSIL, and 38%–91.3% of SCC cases [
8,
9,
13]. In the present study, grade 3 expression was observed in 66.6% of HSIL cases and 97.4% of SCC cases. These findings demonstrate a trend toward increasing p16 expression grade with lesion severity, reinforcing the utility of p16 as a biomarker of cervical lesion progression and malignancy.
LSIL lesions predominantly exhibit lower p16 expression grades, whereas HSIL and SCC show higher grades, with a progressive increase in staining intensity from LSIL to SCC [
8,
9,
13,
16,
18,
19,
29]. In the present study, a similar progressive increase in p16 intensity was observed, ranging from predominantly negative or weak staining in CIN 1 to predominantly strong staining in SCC. Reported frequencies of weak, moderate, and strong p16 intensity range from 12.5%–50% for CIN 1/LSIL, 0%–77.8% for HSIL (CIN 2 and CIN 3), and 60%–100% for SCC [
8,
11,
18,
19]. Khamseh et al. [
18] reported 87.5% negative staining in CIN 1, while 77.8% of CIN 3 cases showed moderate intensity. Li et al. [
11] reported strong p16 intensity in 60% of SCC cases. Diouf et al. [
19] observed 100% strong staining in SCC, with lower intensities in LSIL (50% weak) and HSIL (42.9% each moderate and strong). Singh et al. [
8] reported LSIL cases with 37.5% weak and moderate staining each, HSIL with 60% strong staining, and SCC with 81% strong staining. In the present study, LSIL showed weak-to-moderate staining (50% each), HSIL showed 66.6% strong staining, and SCC showed 79.5% strong staining. Collectively, these findings underscore the value of p16 grading and intensity in distinguishing low-grade from high-grade cervical squamous lesions.
Ki-67 expression showed a gradual increase with lesion severity in the present study, consistent with previous reports, supporting its role as a valuable adjunct marker in the diagnosis and grading of cervical squamous lesions and in distinguishing high-grade lesions [
8,
9,
16,
19,
21,
22,
25,
30]. CIN 1/LSIL lesions predominantly show low Ki-67 scores (0–1), with most cases having <25% positive cells [
8,
9,
16,
19,
21]. CIN 2 and CIN 3, or HSIL lesions, demonstrate progressively increased Ki-67 expression, with CIN 3 showing the highest levels [
8,
9,
16,
19,
21]. Zhong et al. [
9] reported that 72.8% of CIN 3 cases exhibited >50% Ki-67 positivity. SCC consistently shows the highest Ki-67 expression, with 64.7%–100% of cases demonstrating >50% positivity across studies [
8,
16,
21]. Diouf et al. [
19] reported >50% Ki-67 positivity in 100% of SCC cases, whereas LSIL cases showed <5% positivity in 50% of cases. The findings of the present study are in concordance with the existing literature, with LSIL showing minimal staining (100% score 0) and SCC demonstrating >50% positivity in 76.9% of cases.
ER expression has been reported to gradually decrease with lesion severity, ranging from 12.5% to 50% in LSIL, 0% to 23.1% in HSIL, and 0% to 38.6% in SCC [
7,
22,
24,
30-
33]. Kanai et al. [
30] demonstrated a progressive decrease in ER positivity from CIN 1 (50%) to CIN 3 (0%). In contrast, Tervahauta et al. [
31] reported ER positivity in 12.5% of CIN 1 and 23.1% of CIN 3 cases. Nikolaou et al. [
22] found ER positivity in 30.6% of CIN cases, while Fonseca-Moutinho et al. [
32] reported ER positivity in 44% of CIN lesions. In SCC, ER expression has been variably reported, ranging from 0% in the study by Mosny et al. [
33] to 38.6% in the study by Sun et al. [
7]. In the present study, ER negativity was observed in 0% of LSIL, 33.3% of HSIL, and 53.8% of SCC cases. This observation may suggest a trend toward decreasing ER expression with increasing lesion severity in this cohort, consistent with earlier reports in which LSIL demonstrated some ER positivity and SCC often showed lower expression. ER expression intensity also decreased with lesion progression in the present study. Higher ER positivity was observed among women aged >45 years, women with early menarche, postmenopausal women, multiparous women, and women with early childbearing age. ER positivity was also slightly higher in non-keratinizing tumors compared with keratinizing tumors and increased with decreasing tumor differentiation. However, no statistically significant correlation was identified between ER expression and the evaluated clinicopathological parameters, similar to findings reported in previous studies [
7,
24,
32].
In published studies, HER2 positivity rates in cervical squamous premalignant and malignant lesions range widely from 0% to 64%, while reported negativity rates range from 8% to 97% [
5,
7,
10,
12,
15,
23,
25-
27,
31,
34-
38]. In the present study, only 5.1% of SCC cases were HER2 positive. Most studies have reported predominantly negative HER2 expression in cervical squamous lesions [
5,
7,
12,
15,
23,
34-
36,
38]. Gupta et al. [
27] and Bajpai et al. [
5] reported higher HER2 positivity in poorly differentiated SCC; however, the single poorly differentiated SCC case in our cohort was HER2 negative. Several studies have also reported no significant correlation between HER2 expression and clinicopathological parameters, findings that are concordant with our study [
23,
36,
39]. In contrast, some authors have demonstrated a significant association between HER2 expression and histological grade, a relationship that was not observed in our cohort [
10,
27]. Additionally, the present study does not show a significant correlation between ER and HER2 in cervical premalignant and malignant lesions, consistent with the findings of Sun et al. [
7].
The limitations of the present study include its restriction to p16-positive cases, which may limit generalizability to the broader spectrum of cervical lesions and reduce statistical power for detecting associations. Variations in immunohistochemistry protocols, scoring systems, and interpretation criteria across studies may also contribute to differences between the present findings and previously published results. Application of these findings in clinical practice will require validation through larger, multi-institutional studies and integration with emerging therapeutic approaches to better define their potential impact on patient outcomes.
In conclusion, the present study demonstrates that p16 positivity increases in both intensity and grade from LSIL to SCC. In most CIN cases, Ki-67 expression was commonly observed up to the middle two-thirds of the dysplastic epithelium. ER expression was observed to decrease with lesion progression; however, this trend did not reach statistical significance in the present study. HER2 expression was rarely positive in cervical squamous neoplasia, suggesting limited therapeutic applicability. Overall, these findings provide valuable insights into the biological behavior of cervical squamous neoplasms and their potential relevance for targeted therapy. The results of the current study may help address gaps in the existing literature and offer foundational evidence to support further exploration of emerging strategies, including ER- and HER2-related therapeutic approaches.
-
Authors’ contribution
Conceptualization: AP, JSN, IP, AR, SP, NK. Data curation: AP, JSN, NK. Formal analysis: AP, JSN, IP, AR, SP, NK. Methodology: AP, JSN, IP, AR, SP, NK. Validation: AP, JSN, IP, AR, SP, NK. Writing–original draft: AP, JSN, IP, AR, SP, NK. Writing–review & editing: AP, JSN, IP, AR, SP, NK.
-
Conflict of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
This published article includes all data generated or analyzed during this study.
-
Acknowledgments
None.
-
Supplementary materials
None.
Fig. 1.(A) Keratinizing squamous cell carcinoma showing tumor cells with keratin pearl formation (hematoxylin and eosin, ×100). (B) Non-keratinizing squamous cell carcinoma showing a moderately differentiated tumor arranged in sheets and nests (hematoxylin and eosin, ×50). (C) p16 immunohistochemistry showing strong nuclear and cytoplasmic block-type positivity in squamous cell carcinoma (Clone JC8, ×100). (D) Ki-67 immunohistochemistry demonstrating a high proliferative index with nuclear positivity in >75% of tumor cells (Clone MIB-1, ×50). (E) Estrogen receptor immunohistochemistry showing strong nuclear positivity in squamous cell carcinoma, a finding that is usually uncommon (Clone EP1, ×50). (F) Human epidermal growth factor receptor 2 immunohistochemistry showing strong, complete membranous expression (score 3), a rare finding observed in only 2 cases in the present study, in squamous cell carcinoma (Clone EP3, ×100).
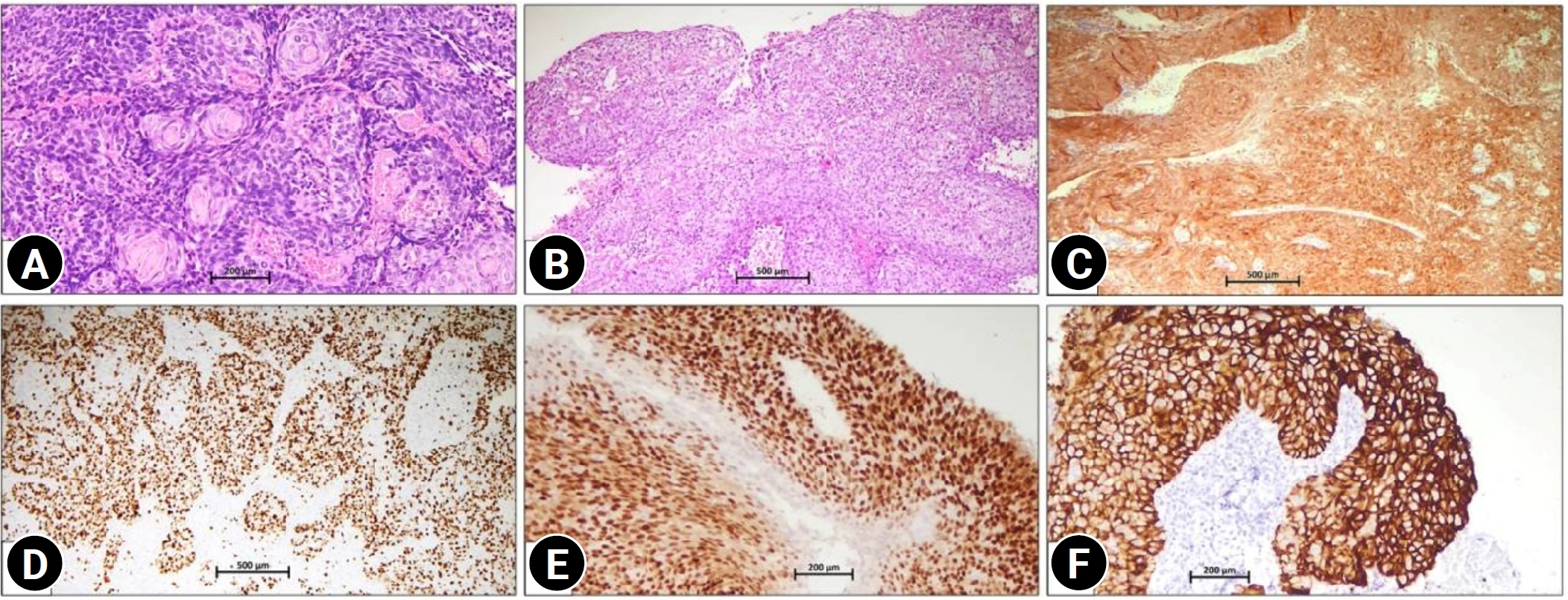
Fig. 2.(A) Estrogen receptor (ER) staining pattern shown as stacked bars representing the proportion of cases with absent, weak, moderate, or strong ER intensity and the corresponding percentage of positive cells (Allred score >3 considered positive). (B) Extent of ER expression in cervical intraepithelial neoplasia cases only (n=17), categorized by epithelial layer involvement as full thickness, middle two-thirds, lower one-third, or absent (29.4%, 5/17).
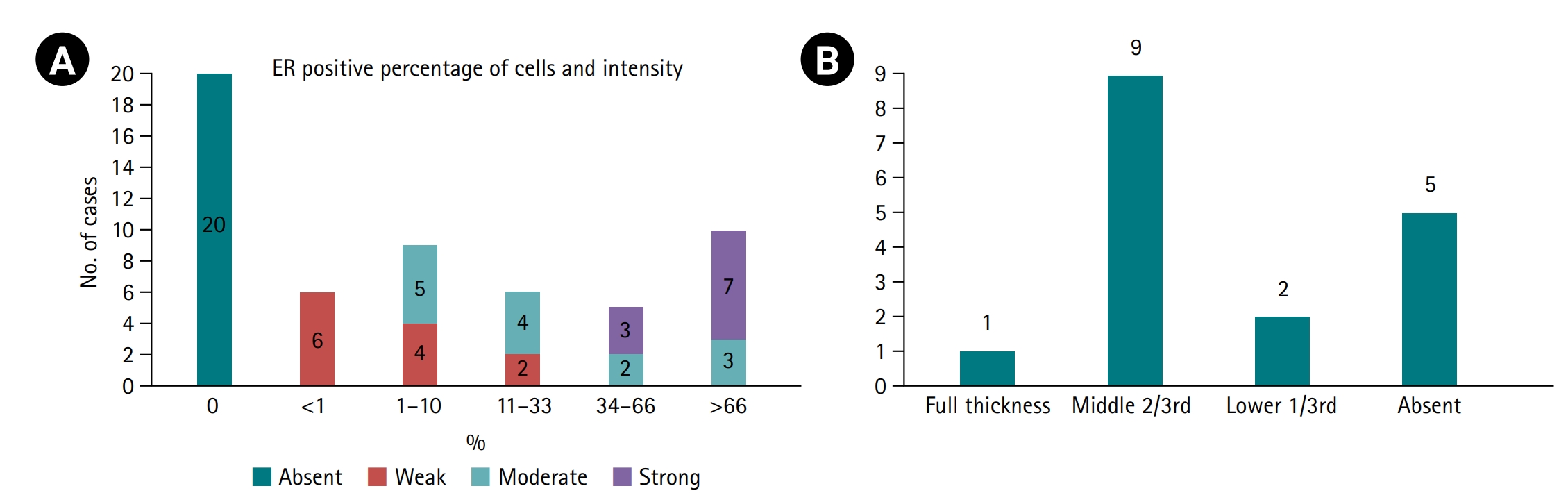
Table 1.Distribution of cervical lesions with clinical and pathological parameters
|
Characteristic |
LSIL |
HSIL |
SCC |
|
Age (yr) |
|
|
|
|
≤45 |
1 |
8 |
7 |
|
>45 |
1 |
7 |
32 |
|
Age at menarche (yr) |
|
|
|
|
≤12 |
1 |
9 |
26 |
|
>12 |
1 |
6 |
13 |
|
Age at first delivery (yr) |
|
|
|
|
≤18 |
1 |
3 |
24 |
|
>18 |
1 |
12 |
15 |
|
Menopausal status |
|
|
|
|
Premenopausal |
2 |
12 |
8 |
|
Postmenopausal |
0 |
3 |
31 |
|
Education status |
|
|
|
|
Illiterate |
0 |
5 |
33 |
|
Literate |
2 |
10 |
6 |
|
Vaginal bleeding |
|
|
|
|
Present |
2 |
13 |
39 |
|
Absent |
0 |
2 |
0 |
|
Pain |
|
|
|
|
Present |
0 |
5 |
29 |
|
Absent |
2 |
10 |
10 |
|
White discharge |
|
|
|
|
Present |
2 |
11 |
26 |
|
Absent |
0 |
4 |
13 |
|
Foul-smelling discharge |
|
|
|
|
Present |
0 |
2 |
8 |
|
Absent |
2 |
13 |
31 |
|
Weight loss |
|
|
|
|
Present |
0 |
1 |
29 |
|
Absent |
2 |
14 |
10 |
|
p16 intensity |
|
|
|
|
Weak |
1 |
0 |
0 |
|
Moderate |
1 |
5 |
8 |
|
Strong |
0 |
10 |
31 |
|
p16 grade |
|
|
|
|
Grade 1 |
2 |
1 |
0 |
|
Grade 2 |
0 |
4 |
1 |
|
Grade 3 |
0 |
10 |
38 |
|
Ki-67 expression |
|
|
|
|
<5% |
2 |
4 |
3 |
|
6%–25% |
0 |
1 |
1 |
|
26%–50% |
0 |
2 |
5 |
|
51%–<75% |
0 |
6 |
17 |
|
>75% |
0 |
2 |
13 |
|
ER intensity in LSIL, HSIL, and SCC |
|
|
|
|
Absent |
0 |
5 |
15 |
|
Weak |
0 |
4 |
8 |
|
Moderate |
0 |
3 |
11 |
|
Strong |
2 |
3 |
5 |
Table 2.Association of ER and HER2 status with clinicopathological variables
|
Variable |
ER-negative |
ER-positive |
P-value |
HER2 status |
P-value |
|
Negative |
Equivocal |
Positive |
|
Age (yr) |
|
|
0.799 |
|
|
|
0.660 |
|
≤45 |
7 |
9 |
|
13 |
2 |
1 |
|
|
>45 |
19 |
21 |
|
31 |
8 |
1 |
|
|
Age at menarche (yr) |
|
|
0.905 |
|
|
|
0.258 |
|
≤12 |
16 |
20 |
|
26 |
8 |
2 |
|
|
>12 |
10 |
10 |
|
18 |
2 |
0 |
|
|
Menopausal status |
|
|
0.906 |
|
|
|
0.948 |
|
Premenopausal |
10 |
12 |
|
17 |
4 |
1 |
|
|
Postmenopausal |
16 |
18 |
|
27 |
6 |
1 |
|
|
Parity |
|
|
0.348 |
|
|
|
0.870 |
|
Multipara |
26 |
29 |
|
43 |
10 |
2 |
|
|
Nullipara |
0 |
1 |
|
1 |
0 |
0 |
|
|
Age at first delivery (yr) |
|
|
1.000 |
|
|
|
1.000 |
|
≤18 |
13 |
15 |
|
22 |
5 |
1 |
|
|
>18 |
13 |
15 |
|
22 |
5 |
1 |
|
|
White discharge |
|
|
0.950 |
|
|
|
0.632 |
|
Absent |
8 |
9 |
|
14 |
3 |
0 |
|
|
Present |
18 |
21 |
|
30 |
7 |
2 |
|
|
Foul-smelling discharge |
|
|
0.250 |
|
|
|
0.791 |
|
Absent |
23 |
23 |
|
36 |
8 |
2 |
|
|
Present |
3 |
7 |
|
8 |
2 |
0 |
|
|
Vaginal bleeding |
|
|
0.180 |
|
|
|
0.754 |
|
Absent |
0 |
2 |
|
2 |
0 |
0 |
|
|
Present |
26 |
28 |
|
42 |
10 |
2 |
|
|
Pain |
|
|
0.906 |
|
|
|
0.410 |
|
Absent |
10 |
12 |
|
17 |
5 |
0 |
|
|
Present |
16 |
18 |
|
27 |
5 |
2 |
|
|
Weight loss |
|
|
0.266 |
|
|
|
0.288 |
|
Absent |
10 |
16 |
|
20 |
6 |
0 |
|
|
Present |
16 |
14 |
|
24 |
4 |
2 |
|
|
Histological diagnosis |
|
|
0.176 |
|
|
|
0.573 |
|
LSIL |
0 |
2 |
|
1 |
1 |
0 |
|
|
HSIL |
5 |
10 |
|
13 |
2 |
0 |
|
|
SCC |
21 |
18 |
|
30 |
7 |
2 |
|
|
Histological type of SCC (39 cases) |
|
|
0.734 |
|
|
|
0.220 |
|
Keratinizing |
8 |
5 |
|
8 |
4 |
1 |
|
|
Non-keratinizing |
13 |
13 |
|
22 |
3 |
1 |
|
|
Histological grade of SCC (39 cases) |
|
|
0.387 |
|
|
|
0.733 |
|
Well-differentiated |
3 |
1 |
|
4 |
0 |
0 |
|
|
Moderately-differentiated |
18 |
16 |
|
25 |
7 |
2 |
|
|
Poorly-differentiated |
0 |
1 |
|
1 |
0 |
0 |
|
|
p16 grade |
|
|
0.099 |
|
|
|
0.932 |
|
Grade 1 |
0 |
3 |
|
2 |
1 |
0 |
|
|
Grade 2 |
1 |
4 |
|
4 |
1 |
0 |
|
|
Grade 3 |
25 |
23 |
|
38 |
8 |
2 |
|
|
p16 intensity |
|
|
0.388 |
|
|
|
0.897 |
|
Weak |
0 |
1 |
|
1 |
0 |
0 |
|
|
Moderate |
5 |
9 |
|
11 |
3 |
0 |
|
|
Strong |
21 |
20 |
|
32 |
7 |
2 |
|
|
Ki-67 in CIN (17 cases) |
|
|
0.575 |
|
|
|
1.000 |
|
Lower 1/3rd |
1 |
4 |
|
4 |
1 |
0 |
|
|
Middle 2/3rd |
2 |
6 |
|
7 |
1 |
0 |
|
|
Full |
2 |
2 |
|
3 |
1 |
0 |
|
|
SCC Ki-67 (39 cases) |
|
|
0.752 |
|
|
|
0.303 |
|
≤60% |
9 |
9 |
|
16 |
2 |
0 |
|
|
>60% |
12 |
9 |
|
14 |
5 |
2 |
|
|
HER2 status |
|
|
0.280 |
|
|
|
|
|
Negative |
19 |
25 |
|
|
|
|
|
|
Equivocal |
5 |
5 |
|
|
|
|
|
|
Positive |
2 |
0 |
|
|
|
|
|
References
- 1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024;74:229-263. https://doi.org/10.3322/caac.21834
- 2. Herrington CS, Kim KR, Kong CS, Longacre TA, McCluggage WG, Mikami Y, Ordi J, Soslow RA. Tumours of the uterine cervix. In: WHO Classification of Tumours Editorial Board, editor. Female genital tumours. International Agency for Research on Cancer; 2020. p. 336-351.
- 3. Pan D, Wei K, Ling Y, Su S, Zhu M, Chen G. The prognostic role of Ki-67/MIB-1 in cervical cancer: a systematic review with meta-analysis. Med Sci Monit 2015;21:882-889. https://doi.org/10.12659/MSM.892807
- 4. Chung SH, Franceschi S, Lambert PF. Estrogen and ERalpha: culprits in cervical cancer? Trends Endocrinol Metab 2010;21:504-511. https://doi.org/10.1016/j.tem.2010.03.005
- 5. Bajpai S, Awasthi S, Dutta S, Mittal A, Kumar A, Ahmad F. Role of HER-2/neu in premalignant and malignant lesions of uterine cervix. J Clin Diagn Res 2017;11:EC01-EC04. https://doi.org/10.7860/JCDR/2017/26583.10547
- 6. Itkin B, Garcia A, Straminsky S, Adelchanow ED, Pereyra M, Haab GA, Bardach A. Prevalence of HER2 overexpression and amplification in cervical cancer: a systematic review and meta-analysis. PLoS One 2021;16:e0257976. https://doi.org/10.1371/journal.pone.0257976
- 7. Sun L, Schroeder MC, Hagemann IS, Pfeifer JD, Schwarz JK, Grigsby PW, Markovina S, Lin AJ. Expression of potential biomarker targets by immunohistochemistry in cervical carcinomas. Int J Gynecol Pathol 2022;41:628-635. https://doi.org/10.1097/PGP.0000000000000853
- 8. Singh P, Kaushik S, Thakur B, Acharya S, Bhardwaj A, Bahal N. Evaluation of variable p16 immunostaining patterns, Ki-67 indices and HPV status in cervical SILs and squamous cell carcinomas: an institutional experience. Indian J Pathol Microbiol 2023;66:63-69. https://doi.org/10.4103/ijpm.ijpm_656_21
- 9. Zhong P, Li J, Gu Y, Liu Y, Wang A, Sun Y, Lu L. P16 and Ki-67 expression improves the diagnostic accuracy of cervical lesions but not predict persistent high risk human papillomavirus infection with CIN1. Int J Clin Exp Pathol 2015;8:2979-2986.
- 10. Varshney S, Maheshwari V, Aijaz M, Alam K. Role and significance of HER-2/neu as a biomarker in the premalignant and malignant lesions of uterine cervix. Ann Diagn Pathol 2020;45:151443. https://doi.org/10.1016/j.anndiagpath.2019.151443
- 11. Li JG, Li L, Zhang SW. Different expression of p16INK4a and p14ARF in cervical and lung cancers. Eur Rev Med Pharmacol Sci 2013;17:3007-3011.
- 12. Conesa-Zamora P, Torres-Moreno D, Isaac MA, Pérez-Guillermo M. Gene amplification and immunohistochemical expression of ERBB2 and EGFR in cervical carcinogenesis: correlation with cell-cycle markers and HPV presence. Exp Mol Pathol 2013;95:151-155. https://doi.org/10.1016/j.yexmp.2013.06.011
- 13. Liu HQ, Wang YH, Wang LL, Hao M. P16INK4A and survivin: diagnostic and prognostic markers in cervical intraepithelial neoplasia and cervical squamous cell carcinoma. Exp Mol Pathol 2015;99:44-49. https://doi.org/10.1016/j.yexmp.2015.04.004
- 14. Tan GC, Sharifah NA, Shiran MS, Salwati S, Hatta AZ, Paul-Ng HO. Utility of Ki-67 and p53 in distinguishing cervical intraepithelial neoplasia 3 from squamous cell carcinoma of the cervix. Asian Pac J Cancer Prev 2008;9:781-784.
- 15. Bhadauria M, Ray A, Grover RK, Sharma S, Naik SL, Sharma BK. Oncoprotein c-erbB-2 in squamous cell carcinoma of the uterine cervix and evaluation of its significance in response of disease to treatment. Indian J Physiol Pharmacol 2001;45:191-198.
- 16. Mehdi HK, Raju K, Sheela SR. Association of P16, Ki-67, and CD44 expression in high-grade squamous intraepithelial neoplasia and squamous cell carcinoma of the cervix. J Cancer Res Ther 2023;19:S260-S267. https://doi.org/10.4103/jcrt.jcrt_43_21
- 17. Kwasniewska A, Postawski K, Gozdzicka-Jozefiak A, Kwasniewski W, Grywalska E, Zdunek M, Korobowicz E. Estrogen and progesterone receptor expression in HPV-positive and HPV-negative cervical carcinomas. Oncol Rep 2011;26:153-160. https://doi.org/10.3892/or.2011.1256
- 18. Khamseh A, Farhadi A, Jalilvand S, Yarandi F, Izadi-Mood N, Ghorbani S, Saadati H, Shirali E, Jazayeri SM, Sarvari J. Analysis of HPV-16 viral load, integration status, and p16 expression in relation to EBV co-infection and cervical lesion severity. Sci Rep 2025;15:8329. https://doi.org/10.1038/s41598-025-93358-x
- 19. Diouf D, Diop G, Fall C, Sarr S, Diarra CA, Ngom AI, Ka S, Lo S, Faye O, Dem A. The association of molecular biomarkers in the diagnosis of cervical pre-cancer and cancer and risk factors in Senegalese. Asian Pac J Cancer Prev 2020;21:3221-3227. https://doi.org/10.31557/APJCP.2020.21.11.3221
- 20. Missaoui N, Trabelsi A, Hmissa S, Fontanière B, Yacoubi MT, Mokni M, Korbi S, Frappart L. P16INK4A overexpression in precancerous and cancerous lesions of the uterine cervix in Tunisian women. Pathol Res Pract 2010;206:550-555. https://doi.org/10.1016/j.prp.2010.02.014
- 21. Yang SF, Yuan SS, Yeh YT, Hung SC, Wu MT, Su JH, Chai CY. Positive association between STAT3 and Ki-67 in cervical intraepithelial neoplasia. Kaohsiung J Med Sci 2006;22:539-546. https://doi.org/10.1016/S1607-551X(09)70350-X
- 22. Nikolaou M, Koumoundourou D, Ravazoula P, Papadopoulou M, Michail G, Decavalas G. An immunohistochemical analysis of sex-steroid receptors, tumor suppressor gene p53 and Ki-67 in the normal and neoplastic uterine cervix squamous epithelium. Med Pregl 2014;67:202-207. https://doi.org/10.2298/mpns1408202n
- 23. Rosty C, Couturier J, Vincent-Salomon A, Genin P, Fréneaux P, Sigal-Zafrani B, Sastre-Garau X. Overexpression/amplification of HER-2/neu is uncommon in invasive carcinoma of the uterine cervix. Int J Gynecol Pathol 2004;23:13-17. https://doi.org/10.1097/01.pgp.0000092137.88121.8d
- 24. Park CS, Joo IS, Song SY, Kim DS, Bae DS, Lee JH. An immunohistochemical analysis of heat shock protein 70, p53, and estrogen receptor status in carcinoma of the uterine cervix. Gynecol Oncol 1999;74:53-60. https://doi.org/10.1006/gyno.1999.5429
- 25. Carreras R, Alameda F, Mancebo G, García-Moreno P, Mariñoso ML, Costa C, Fusté P, Baró T, Serrano S. A study of Ki-67, c-erbB2 and cyclin D-1 expression in CIN-I, CIN-III and squamous cell carcinoma of the cervix. Histol Histopathol 2007;22:587-592. https://doi.org/10.14670/HH-22.587
- 26. Protrka Z, Mitrović S, Arsenijević N, Baskić D, Radosavljević G, Stanković M, Arsenijević S. HER-2 expression in uterine cervix carcinogenesis. J BUON 2007;12:91-97.
- 27. Gupta N, Singh S, Marwah N, Kumar S, Chabra S, Sen R. HER-2/neu expression in lesions of uterine cervix: is it reliable and consistent? Indian J Pathol Microbiol 2009;52:482-485. https://doi.org/10.4103/0377-4929.56127
- 28. Hanprasertpong J, Tungsinmunkong K, Chichareon S, Wootipoom V, Geater A, Buhachat R, Boonyapipat S. Correlation of p53 and Ki-67 (MIB-1) expressions with clinicopathological features and prognosis of early stage cervical squamous cell carcinomas. J Obstet Gynaecol Res 2010;36:572-580. https://doi.org/10.1111/j.1447-0756.2010.01227.x
- 29. Ahmad A, Raish M, Shahid M, Batra S, Batra V, Husain SA. The synergic effect of HPV infection and epigenetic anomaly of the p16 gene in the development of cervical cancer. Cancer Biomark 2017;19:375-381. https://doi.org/10.3233/CBM-160060
- 30. Kanai M, Shiozawa T, Xin L, Nikaido T, Fujii S. Immunohistochemical detection of sex steroid receptors, cyclins, and cyclin-dependent kinases in the normal and neoplastic squamous epithelia of the uterine cervix. Cancer 1998;82:1709-1719. https://doi.org/10.1002/(sici)1097-0142(19980501)82:9<1709::aid-cncr18>3.0.co;2-8
- 31. Tervahauta A, Syrjänen S, Syrjänen K. Epidermal growth factor receptor, c-erbB-2 proto-oncogene and estrogen receptor expression in human papillomavirus lesions of the uterine cervix. Int J Gynecol Pathol 1994;13:234-240. https://doi.org/10.1097/00004347-199407000-00007
- 32. Fonseca-Moutinho JA, Cruz E, Carvalho L, Prazeres HJ, de Lacerda MM, da Silva DP, Mota F, de Oliveira CF. Estrogen receptor, progesterone receptor, and bcl-2 are markers with prognostic significance in CIN III. Int J Gynecol Cancer 2004;14:911-920. https://doi.org/10.1111/j.1048-891X.2004.14529.x
- 33. Mosny DS, Herholz J, Degen W, Bender HG. Immunohistochemical investigations of steroid receptors in normal and neoplastic squamous epithelium of the uterine cervix. Gynecol Oncol 1989;35:373-377. https://doi.org/10.1016/0090-8258(89)90082-6
- 34. Lesnikova I, Lidang M, Hamilton-Dutoit S, Koch J. HER2/neu (c-erbB-2) gene amplification and protein expression are rare in uterine cervical neoplasia: a tissue microarray study of 814 archival specimens. APMIS 2009;117:737-745. https://doi.org/10.1111/j.1600-0463.2009.02531.x
- 35. Brumm C, Rivière A, Wilckens C, Löning T. Immunohistochemical investigation and northern blot analysis of c-erB-2 expression in normal, premalignant and malignant tissues of the corpus and cervix uteri. Virchows Arch A Pathol Anat Histopathol 1990;417:477-484. https://doi.org/10.1007/BF01625727
- 36. Califano D, Losito S, Pisano C, Santelli G, Greggi S, Iodice F, DiVagno G, Silvestro G, Tambaro R, Formato R, Iaffaioli VR, Di Maio M, Pignata S. Significance of erb-B2 immunoreactivity in cervical cancer. Front Biosci 2006;11:2071-2076. https://doi.org/10.2741/1949
- 37. Halle MK, Ojesina AI, Engerud H, Woie K, Tangen IL, Holst F, Høivik E, Kusonmano K, Haldorsen IS, Vintermyr OK, Trovik J, Bertelsen BI, Salvesen HB, Krakstad C. Clinicopathologic and molecular markers in cervical carcinoma: a prospective cohort study. Am J Obstet Gynecol 2017;217:432. https://doi.org/10.1016/j.ajog.2017.05.068
- 38. Kristensen GB, Holm R, Abeler VM, Tropé CG. Evaluation of the prognostic significance of cathepsin D, epidermal growth factor receptor, and c-erbB-2 in early cervical squamous cell carcinoma: an immunohistochemical study. Cancer 1996;78:433-440. https://doi.org/10.1002/(SICI)1097-0142(19960801)78:3<433::AID-CNCR9>3.0.CO;2-K
- 39. Mandai M, Konishi I, Koshiyama M, Komatsu T, Yamamoto S, Nanbu K, Mori T, Fukumoto M. Altered expression of nm23-H1 and c-erbB-2 proteins have prognostic significance in adenocarcinoma but not in squamous cell carcinoma of the uterine cervix. Cancer 1995;75:2523-2529. https://doi.org/10.1002/1097-0142(19950515)75:10<2523::aid-cncr2820751019>3.0.co;2-l
 , Jitendra Singh Nigam1,*
, Jitendra Singh Nigam1,* , Immanuel Pradeep1
, Immanuel Pradeep1 , Ashutosh Rath1
, Ashutosh Rath1 , Seetu Palo1
, Seetu Palo1 , Naina Kumar2
, Naina Kumar2